NJAIChE is proud to continue its service to the AIChE community by sponsoring:
RUTGERS
STUDENT AWARDS
Senior Research Scholar
Senior Leader Scholar
Junior Research Scholar
Junior Leader Scholar
Please join us to network with and support our future colleagues pursuing their degrees at Rutgers. We will be honoring those students who have set and example to their peers. Support the future of chemical engineering at a fun social event.
Meeting Logistics
Agenda:
Networking begins 6:00 PM
Light Dinner and Awards 7:00 PM
All members, or those interested in becoming members, of NJAIChE are welcome. We are interested in your ideas of how to serve Chemical Engineers (present, future, and past) better. The industrial sponsor for this meeting is SSL.
We look forward to seeing you at Rutgers or contact us at https://njaiche.org/contact/ to share your ideas or offer to sponsor a meeting.
See location details at the end of the message
Location:
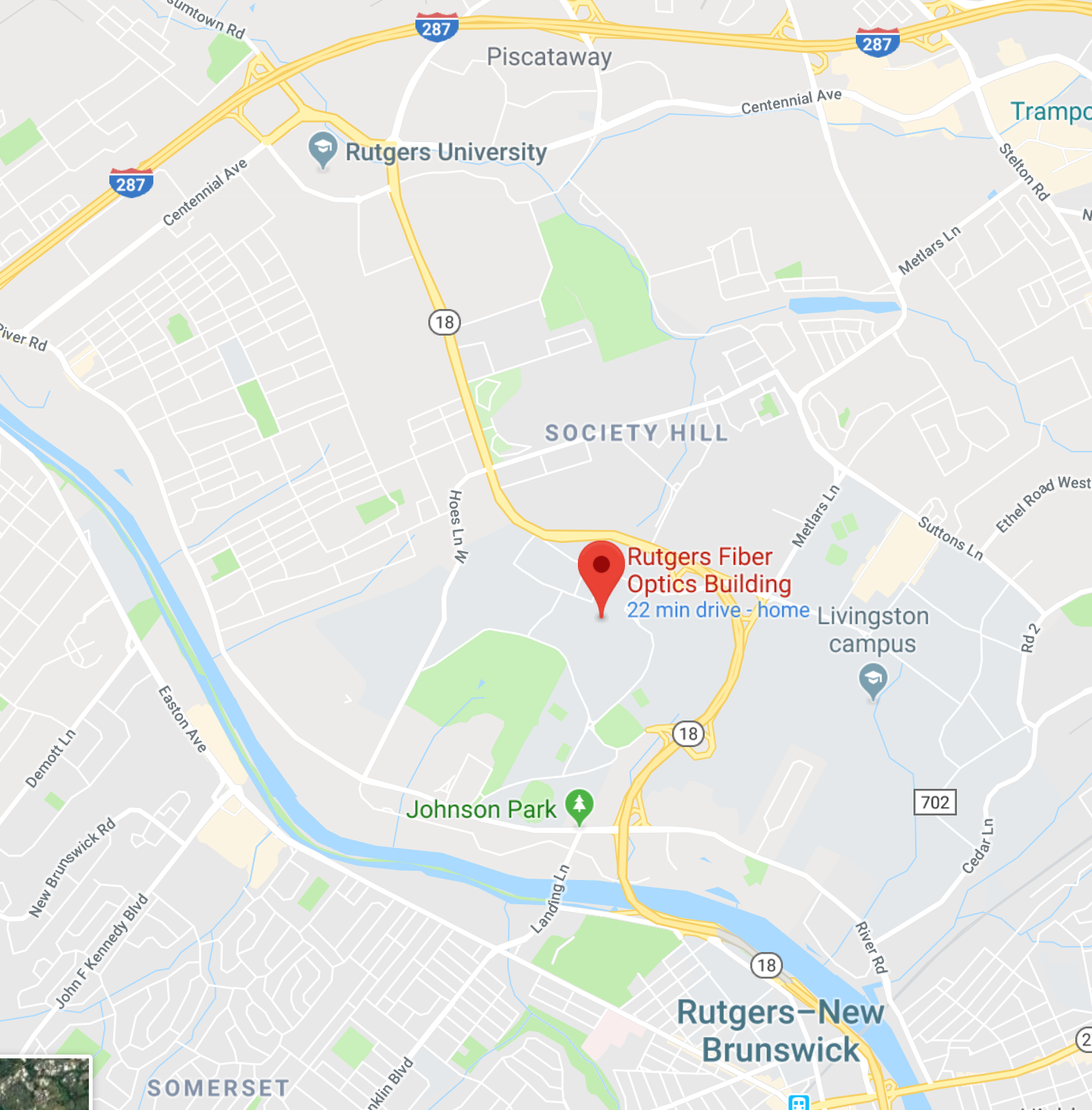
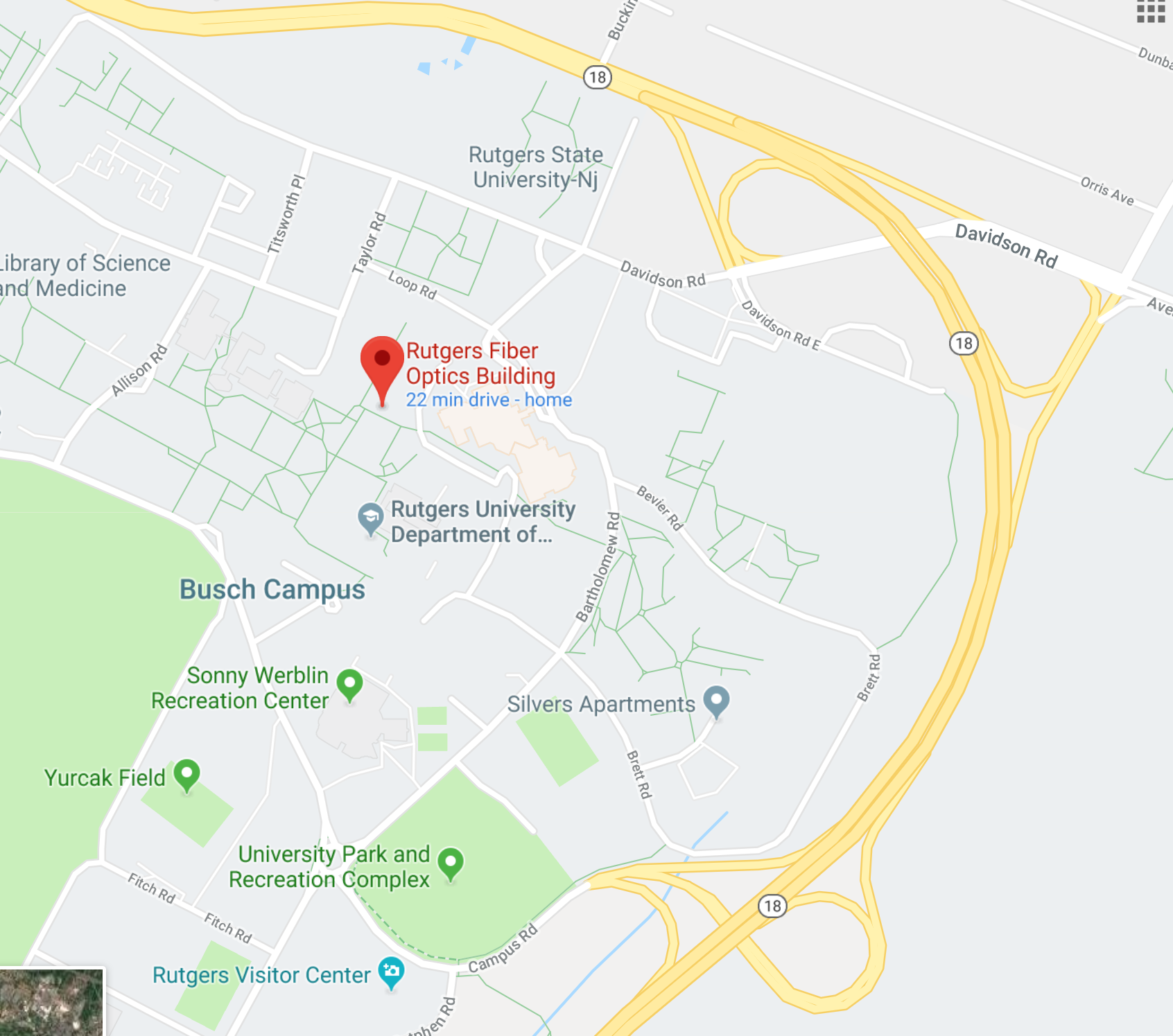
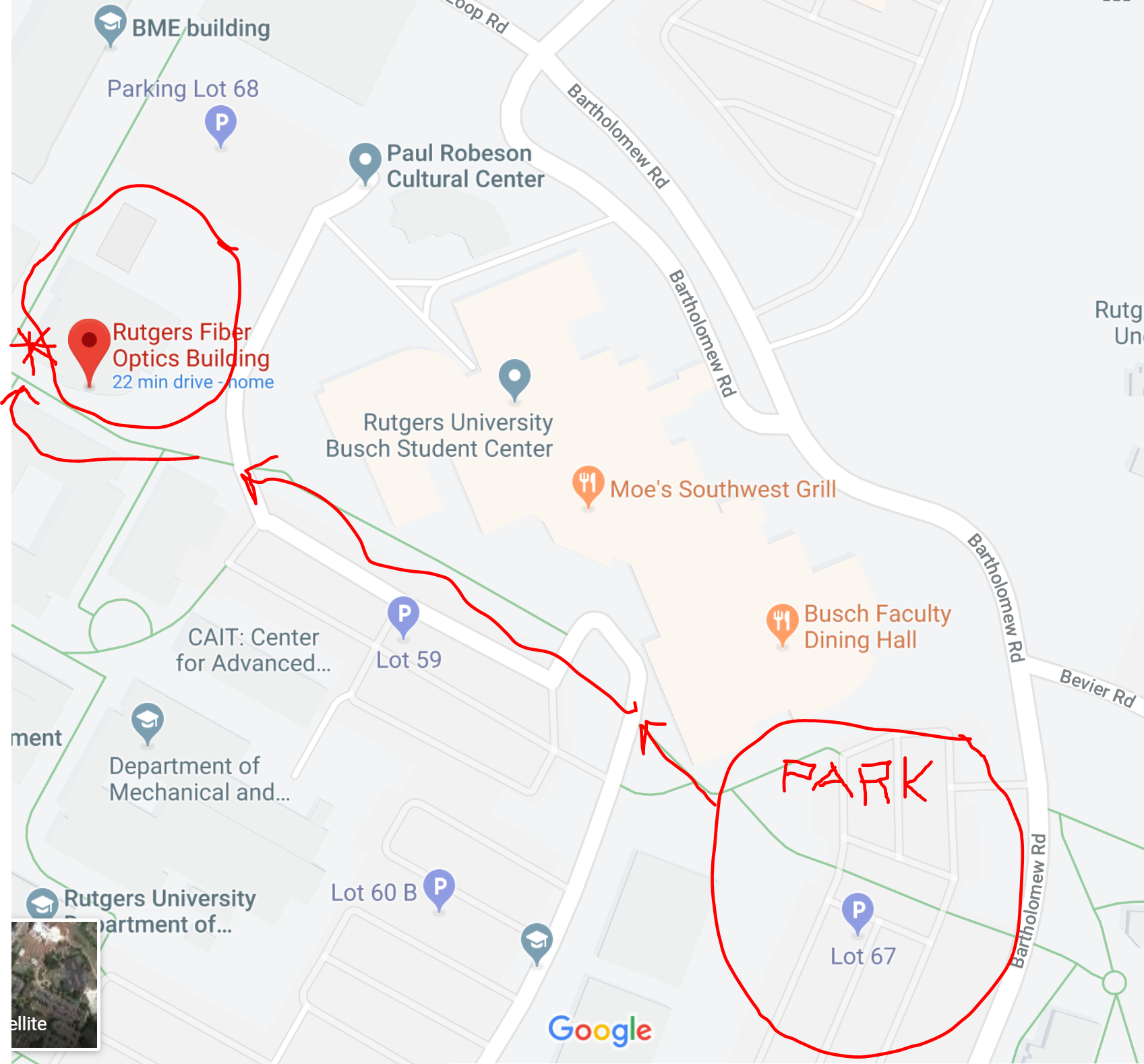
Rutgers University
Elmer C. Easton Engineering Student Hub,
Fiber Optics building,
Busch campus
see maps at end of message
MAPS for RUTGERS